I. Use of Dental Cements
– Each use of dental cement requires specific characteristics.
– Luting materials need to be very fluid when being used to cement a crown and
should become very strong after they set.
– Other cements are mixed to a putty like consistency to restore missing tooth structure or to help protect the pulp under a filling.
– Some cements are used for a variety of procedures; others have limited use.
A. Luting Agent
– The term “cement” implies that the material will be used to lute or glue things together.
– Although other uses are common, use as a luting agent has given this group of
materials their name, cements.
– Dental cements hold appliances and restorations in place with micromechanical and macro mechanical retention.
– Some dental cements are adhesive through chemical bonds, but most are not.
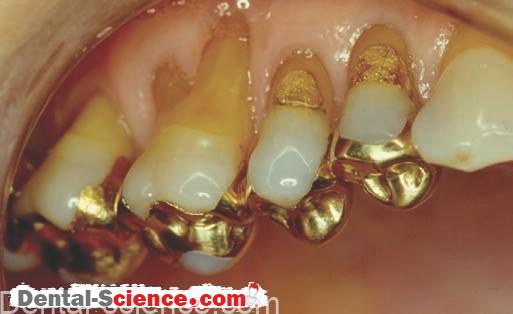
– When a crown is luted to the preparation, the cement is mixed and then painted inside the crown (or filled).
– The crown is seated in place on the preparation, causing the excess cement to ooze out at the margins.
– The cement is allowed to set either partially or completely, and the excess is
removed, much like scaling calculus from teeth.
– It is critical that all the excess cement is removed because any excess cement left
behind will cause gingival irritation.
– Luting cements have the most demanding requirements of any dental material.
– They must set in the mouth, changing from a fluid liquid to a rock-hard solid in
a matter of minutes.
– The resulting material must be biocompatible and insoluble in oral fluids.
– Because cements are much more soluble than the overlying restorative material,
accurate fit of the restoration to be luted is critical.
– Crown margins should precisely fit the preparation to minimize the amount of cement that is exposed to the oral environment.
– Poorly fitting margins increase the solubility of the cement and the likelihood of
recurrent decay.
– A patient’s oral hygiene and diet can also affect the longevity of a luted restoration.
– As bacteria ferment sugars, the pH in the mouth drops.
– Dental cements are much more soluble in an acidic environment.
– As the cement dissolves, a gap is created between the tooth and the restoration.
– The tooth structure at the gap will have a high risk for caries.
– Plaque control and proper diet will reduce the probability of recurrent decay for
a variety of reasons, including maintenance of a less destructive environment for
dental cements and other materials.
B. Pulp Protection
– Dental cements are also used as an intermediate base or liner when the remaining dentin is believed to be less than 2 mm thick.
– A base or liner is placed on the dentin between the pulp and the restorative material.
– Because the solubility of dental cements is much greater than that of the
overlying restorative material, bases and liners must not be placed on margins.
1. Liners
– A liner is used to protect the pulp from chemical irritation.
– A liner may stimulate secondary dentin formation or release fluoride.
– Liners are too thin (0.5 mm) to provide thermal insulation, and they may be too weak to support the restorative material or to resist amalgam condensation forces.
– Several lining materials were originally used as a paint-on barrier composed of a
solvent and calcium hydroxide or zinc oxide.
– Methylcellulose was commonly added to increase viscosity and improve handling.
– Liners were used to prevent chemical irritation.
– Originally, they were very weak and soluble, and they were frequently covered with a stronger base material, such as zinc phosphate cement.
– Liners were also used under composites, when it was believed that the
composites themselves were irritating.
– Dentistry has learned that composites are not irritating and the use of liners has
dramatically decreased.
2. Bases
– A base is stronger and thicker than a liner.
– A base provides thermal insulation.
– Some support the restorative material and may release fluoride, and some are
irritating to the pulp before the setting reaction has completed.
– Such a base may be used in conjunction with a liner.
– Previously, the distinction between bases and liners was clear.
– Today, liner materials are much stronger, and the distinction between bases and
liners is quite blurred.
C. Temporary Restoration
– Some of the same dental cements that are used to lute crowns and that serve as a base may also be used as a temporary restorative material.
– For some cements, the material is mixed to a thicker consistency than is used for
luting.
– Other cements have formulations that are designed for use as temporary and
permanent restorations.
1. Temporary Restorative Materials
– Many dental cements are used as a temporary restorative material.
– The specific cement is chosen based on the particular clinical requirements of the
situation.
– A temporary restoration (filling) may be placed as an emergency procedure when time
restraints prevent a more complex treatment.
– Also, when pulpal pain and other symptoms do not result in a definitive diagnosis, such as reversible versus irreversible pulpitis, a temporary restoration might be placed.
2. Temporary Material as a Base
– At times, a temporary filling is placed, and at a later appointment, part of the
temporary filling material is removed.
– The remaining temporary material then becomes a base, which is covered with
a permanent restoration.
-The advantage to such procedures is that the pulp is less irritated when the overlying dentin is not exposed a second time.
3. Caries Control
– When patients have a high number of carious lesions , caries control procedures may be implemented.
– The goal of caries control is to change the oral environment from cariogenic to
non-cariogenic.
– It is hoped that caries control procedures change the oral flora from acid-producing and acid-loving bacteria to other nonpathogenic species.
– Caries control includes quick, efficient removal of as much decay as possible in the shortest time possible, placement of temporary restorations, diet improvement,
and fluoride supplements.
– Two dental cements that are frequently used for caries control are zinc oxide–eugenol (ZOE) and glass ionomer cement.
– If all the decay is removed from a lesion, the temporary restoration can later function as a base, as previously described.
– Light-cure glass ionomer materials will bond to composite materials to a certain
extent.
– Light-cure glass ionomers may be used for caries control and then veneered with composite material to improve esthetics and surface roughness.
– The ZOE materials are snow white in appearance and, for many patients, are not
suitable for use in the anterior teeth.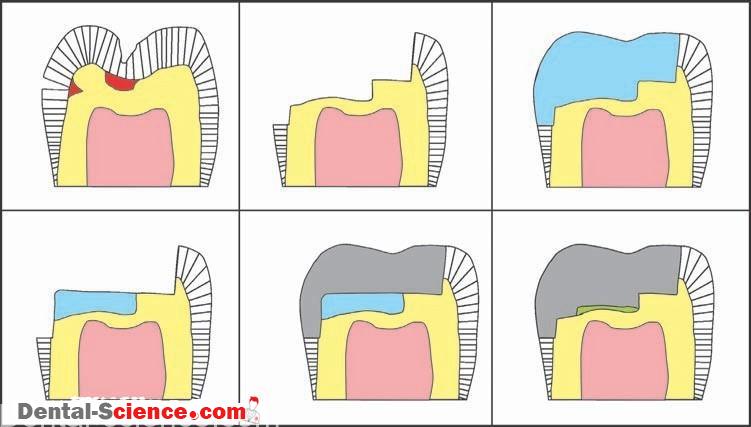
D. Other Formulations of Dental Cements
– Formulations of dental cements are also used as endodontic sealers and
surgical periodontal packs.
– Still other cement formulations include impression materials and bite-registration
materials.
E. Cavity Sealers
– Cavity sealers and dental cements are used to protect the pulp.
1. Varnish
– In dentistry, copal varnish and other varnish formulations are used much like
varnishes are used to protect wood.
– Varnish is composed of resins dissolved in a solvent.
– The varnish is painted onto the entire cavity preparation, including the margins.
– The solvent then evaporates and leaves behind a very thin layer of resin.
– Varnish is frequently used under amalgam restorations.
– It can also function as a chemical barrier, protecting the pulp from an irritating base or
a luting cement.
– It decreases the initial microleakage of the amalgam restoration until corrosion
products form at the interface.
-Varnish is not thick enough to affect thermal sensitivity, and it is not used with
composite materials because it would interfere with adhesion.
2. Dentinal Bonding Agents
– Some dentists are substituting dentinal bonding agents for varnish.
– Several of these products will bond the amalgam to the tooth structure.
– Clinical studies of postoperative sensitivity, however, do not support the routine use of
dentinal bonding systems with all amalgam restorations.
– Some dentists are using Gluma primer as a substitute for varnish.
– Gluma is a brand name for one of the original dentinal bonding systems.
– The original Gluma system has been completely reformulated, but the original primer is still available and becoming more popular with time.
3. Order of Placement
– If more than one base, liner, varnish, or dentinal bonding system is used,
the properties of the materials determine their order of placement.
– Adhesive materials are placed first. Irritating materials are placed last. For example, when a calcium hydroxide liner is used, it is first covered with the varnish, and then the amalgam is placed and condensed.
II. Chemistry of Dental Cements
– With the exception of composite cements, dental cements are brittle,
ceramic materials.
– For many cements, the chemistry is a simple acid–base reaction.
– The resulting product is insoluble in water and oral fluids.
A. Formulation
1. Dental cements are often a powderliquid system.
2. The liquid is an acid.
3. The powder is a base. The powder must be insoluble in oral fluids but reactive with the acid.
4. If one understands the properties of the components of a dental cement, then one will be able to predict the properties of the resulting set material.
B. The Reaction
1. The reaction is acid excess base >>> residual base insoluble salt
2. In terms of the components of the dental cement, the reaction is liquid excess
powder >>> residual powder matrix
3. The residual powder and the matrix must be insoluble in oral fluids.
4. The end result is a “cored structure,” much like that of set amalgam.
C. Composite Cements
– The chemistry of composite cements is the same as that of acrylics and composites.
– This chemistry was presented in Direct Polymeric Restorative Materials.
III. Powders Used in Dental Cements
– Two materials are used to make powders for dental cements: zinc oxide and glass.
-The manufacturing process grinds and sieves the powders to obtain the proper particle size.
– The size of the particles determines the film thickness of the resulting mixed cements.
-Film thickness determines how well a casting or other restoration can be seated on
a preparation.
-Excessively large particles result in high film thickness, open margins, and recurrent decay.
A. Zinc Oxide
1.
– Zinc oxide is the only insoluble, nontoxic, reactive oxide or hydroxide that is available to react with an acid.
– Common additives to the zinc oxide powder are aluminum oxide (alumina) and
other oxides that control the setting rate, such as magnesium oxide.
– Aluminum oxide is very strong and, chemically, very inert.
– It mechanically reinforces the resulting cement.
2. Zinc oxide has some antibacterial effects and is included in diaper rash, sunscreen, and foot powder products.
B. Powdered Glass
1. Silicon oxide, the chemical formula of glass, is very unreactive.
However, if oxides of sodium, calcium, and potassium are added in sufficient
quantity, the glass will react with a strong acid.
As expected, the powder is white because it is made of small, translucent glass
particles.
2. The glass formulation also contains fluoride.
Fluoride is a common glass additive because it reduces the melting temperature and
improves the flow of the molten glass.
Fluoride in the glass powder gives the resulting dental cement the ability to release
fluoride and inhibit recurrent caries.
C. Reactivity of Powders
– The reactivity of the powder components is controlled by the manufacturer and is matched to the reactivity of the liquid component.
IV. Liquids Used in Dental Cements
– The composition or strength of the acid determines the reactivity of the cement liquid.
– The manufacturer controls this.
A. Eugenol
– Eugenol is an organic liquid that is also a weak acid.
– Eugenol is a major component of oil of cloves.
– As a result, eugenol has the distinctive smell and taste (of cloves).
1. Eugenol is a phenol derivative that is antibacterial and also obtundent to the pulp.
Obtundent means that it reduces irritation.
2. Eugenol inhibits free radical polymerization.
This limits the use of eugenol-containing cements because they will inhibit the
set of composite restorative materials.
3. Other organic liquids have been added to eugenol to formulate dental cements.
The most notable of these added organic liquids is ethoxy-benzoic acid.
B. Phosphoric Acid
– The phosphoric acid used in dental cements is approximately two-thirds phosphoric acid and onethird water by weight.
– This formulation is very acidic and can be quite irritating to biologic tissues in or out of the oral cavity.
-The amount of water present affects the reactivity of the liquid by changing the
ionization of phosphoric acid.
-Therefore, it is important to keep the cap on the bottle and not dispense the liquid until one is ready to mix the cement.
– High or low humidity will affect the water content and, therefore, the pH, reactivity, and properties of the resulting cement. If the liquid appears cloudy, it has outlived its shelf-life and should be discarded.
C. Polyacrylic Acid
– Several dental cements use an aqueous solution of polyacrylic acid.
– These solutions are 30% to 50% polyacrylic acid by weight and are very viscous
liquids.
1. Dispensing
– Dispensing these liquids requires more attention than dispensing other cement
liquids.
– If one is not careful, the viscous liquid does not form independent drops.
– Instead, the drops can “run together,” and the amount of liquid dispensed will
not be accurate and will vary greatly with each mix.
– Like phosphoric acid, the liquid should not be dispensed until one is ready to mix the cement because water can evaporate, changing the reactivity and the cement
properties.
– These cement liquids should not be stored in the refrigerator because some will gel and
become unusable.
2. Bonding
– The carboxyl groups of polyacrylic acid bond to calcium in tooth structure.
– This bond is believed to be relatively stable in a wet environment.
– Adhesion of glass ionomer materials was presented in Adhesive Materials.
3. Water Hardening Cements
– “Water hardening” or “water setting” cements use an hydrous, freeze-dried polyacrylic acid.
– The manufacturer mixes zinc oxide or glass powder with the powdered
an hydrous polyacrylic acid.
– This combined powder is mixed with a companion liquid that is predominately water.
– When mixed, the polyacrylic acid first dissolves in the water and then reacts with the
zinc oxide or powdered glass.
V. Powder/Liquid Ratios and Systems of Dental Cements
– Dental cements combine these three liquids and two powders.
– NextTable lists the resulting cements.
Components and the Resulting Cements | ||
Components | Zinc Oxide Powder | Glass Powder |
Eugenol | Zinc oxide–eugenol (ZOE) cement | No reaction |
Phosphoric acid | Zinc phosphate cement | Silicate cement |
Polyacrylic acid | Polycarboxylate cement | Glass ionomer cement |
– The properties of the resulting cements are based on the properties of the
components involved.
– The manufacturer adjusts the reactivity of both the liquid and powder components to obtain proper setting characteristics and other properties.
– Do not mix powders and liquids of different cements or different products of the same type.
A. Components Dictate Handling and Mixing
1.
– The ZOE cement and zinc phosphate cement are mixed with a powder liquid ratio
that depends on the intended use.
– A base or temporary restoration mix is thicker than a luting mix.
– The higher the powder liquid ratio, the greater the strength, the lower the solubility, and in general, the better the cement.
– On the other hand, working time decreases, and viscosity increases.
– If a luting mix becomes too thick, the restoration may not seat adequately.
– In this case, the marginal gap is increased, as is the likelihood of caries.
– In addition, micromechanical retention is reduced.
2. The powder liquid ratio is limited in that the liquid must wet all the powder for the
cement to function adequately.
3.
– Glass ionomer and polycarboxylate cements have powder liquid ratios that
are determined by the manufacturer; it is important to follow the
manufacturer’s directions.
– Mixing time is also important.
– If the mixing procedure is too slow, two problems can occur.
– The first involves the resulting mix becoming too thick. In this case, the restoration may not adequately seat when luted, which increases the marginal gap.
– The second involves adhesion.
– The mixed cement must be fluid enough to wet the tooth for both micromechanical and chemical adhesion.
– If the mixing process is too slow, the carboxylic acid groups react with the powder and are not available to react with the tooth structure.
– Chemical adhesion is then reduced or even eliminated.
4.
– Paper pads are available for mixing many dental cements and other dental materials.
– One must be careful, however, if a paper-mixing pad is used for mixing dental
cement.
– Cement liquids may weaken the paper surface, causing it to become abraded.
– The abraded paper particles are then incorporated into and weaken the resulting
cement.
– Some mixing pads use paper that has been coated with a thin layer of plastic.
– This type of pad is commonly used with glass ionomer and polycarboxylate cements.
– A thick glass slab is favored for mixing zinc phosphate cement; typically, the slab is cooled to improve the resulting mix.
B. Systems
– Most cements come as powder liquid systems, but some are paste paste systems.
– Paste paste systems are dispensed by equal lengths, as are other paste paste
dental materials.
– Each dental cement has its own advantages and disadvantages.
– Use and mixing of dental cements are summarized in Next table.
Reference Guide for Liners, Bases, and Cements | |||||
Cement |
Use |
Mixing Technique |
Mixing Time | Characteristics of a Proper Mix |
Setting Time |
Calcium hydroxide |
Cavity liner | Mix quickly in |
10 seconds |
Uniform color |
2–3 minutes |
Zinc phosphate |
Luting agent | Add divided increments in specified time, using large slab area |
1–1.5 minutes | Mix will stretch 1 inch between slab and spatula |
5.5 minutes |
Base | Same as luting agent (above) |
1–1.5 minutes | Thick, putty-like (non sticking) consistency |
5.5 minutes | |
Glass ionomer |
Luting agent | Add powder to liquid in one portion |
30–45 seconds |
Use while glossy |
7 minutes |
| Paste/paste; mix equal amounts |
20–30 seconds |
Uniform color |
5 minutes | |
Base | Same as luting agent (above) |
30–45 seconds |
Use while glossy |
7 minutes | |
Zinc oxide eugenol (ZOE) | Base and temporary restoration | Add half-scoop increments, using small mixing area |
1.5 minutes | Thick, putty-like (almost crumbly) consistency |
2.5–3.5 minutes |
Polycarboxylate | Luting agent and intermediate base | Add powder to liquid in one portion |
30 seconds |
Use while glossy |
10 minutes |
Temporary | Temporary luting agent | Equal lengths; mix all at once |
30 seconds |
Uniform color |
5 minutes |
Composite |
Luting agent | Mix equal amounts of pastes |
30–45 seconds |
Uniform color |
5 minutes |
These are approximate time periods. Check the manufacturer’s instructions for specific mixing times, measurements, and setting times. | |||||
VI. Types of dental cements
– There are different types of cements as follow:
1. ZOE
2. Zinc phosphate
3. Glass ionomer
4. Polycarboxylate
5. Composite
6. Others
1. ZOE Cement
– Check this link :
2. Zinc phosphate Cement
– Check this link :
3. Glass ionomer Cement
– Check this link :
4. Polycarboxylate Cement
– Check this link :
5. Composite Cement
– Check this link :
6. Others Cements
A. Silicate Cement
– Silicate cement is formed when a glass powder is mixed with phosphoric acid.
– Silicate cement is an old material and is not used today; however, a short discussion is
included for historical perspective.
– At one time, silicate cement was the only available esthetic anterior restorative material.
– Silicate cement is very acidic, very irritating to the pulp, and highly soluble.
– It also leaks excessively.
– As a result, silicate cements were used with liners.
– Silicate cement restorations needed to be replaced often; however, recurrent decay was rare.
– It was discovered that the infrequent occurrence of recurrent decay was caused by fluoride release.
– Fluoride was present in the glass powder. Thus, fluoride release became a desirable property for many dental materials.
B. Calcium Hydroxide Liners and Bases
– At one time, calcium hydroxide liners and bases were very popular materials and were placed under most composite restorations.
– Calcium hydroxide products promote the formation of secondary dentin.
– They are still recommended when a cavity preparation leaves little dentin covering the pulp or when a “micro pulp exposure” is suspected.
– At this time, they are the recommended materials for direct pulp-capping procedures.
– A direct pulp cap is a material that is placed on vital pulp tissue when the overlying dentin is removed and the pulp is exposed.
– The tooth is then restored without root canal therapy.
– Success of a direct pulp cap is not guaranteed, and additional treatment may be required.
– Calcium hydroxide materials are paste/paste systems, as shown in.
– One paste contains calcium hydroxide; the other contains salicylate.
– Salicylate is a weak acid that is chemically similar to eugenol and reacts with the calcium
hydroxide.
– Titanium oxide is added as a filler.
– The oldest calcium hydroxide products were weak, soluble, and used only as liners.
– Improvements have strengthened the material, however, and now some products are strong enough to be used as a base.
– With the development of dentinal bonding systems and our current understanding of the
biocompatibility of composite materials, use of calcium hydroxide products has greatly
diminished.
– The setting reaction of calcium hydroxide materials is accelerated by water.
– The moisture in dentin is sufficient to cause the material to set within seconds of its
application onto the dentin surface.
– This feature makes calcium hydroxide very easy to use.
C. Temporary Cements
– Temporary cements are used to retain a temporary restoration while the permanent
restoration is being fabricated.
– Temporary cements are typically paste/paste systems.
– They are a unique group of materials in that both a maximum and a minimum strength are required (remember Goldilocks?).
– If the temporary cement is too weak, the temporary restoration will be lost prematurely.
– If the temporary cement is too strong, the dentist may have trouble removing the temporary crown, and damage to tissues may occur.
– Many temporary cements are paste/paste ZOE formulations.
– The composition of one paste is zinc oxide and vegetable oil.
– The other paste contains eugenol.
– The ZOE temporary cements mix easily and set quickly in the humidity of the mouth.
– The set material is brittle, and the excess cement is easily removed.
– The obtundent property may even settle down an irritated pulp after it has been insulted by tooth preparation, impression procedures, and construction of a temporary crown.
– This describes a nearly ideal material—so what’s the problem?
– With the development and use of resin cements, many dentists believe that using ZOE
temporary cement will inhibit the set of resin cement.
– Remember that eugenol inhibits free radical polymerization.
– As a result, temporary cements were developed that do not contain eugenol; instead, fatty acids and other chemicals are used to react with zinc oxide.
– These products have similar properties, but they do not handle as well.
– Many are not brittle. Rather, they are “gummy,” and the excess cement can be difficult to
remove.
– At times, a temporary crown or bridge refuses to stay in place.
– It keeps coming off, frustrating both the dentist and the patient.
– When a stronger temporary cement is needed, a powder/liquid formulation of ZOE cement can be used but should be mixed with a lower-than-recommended powder/liquid ratio to reduce the strength.
D. Surgical/Periodontal Packs
– Surgical periodontal packs are paste/paste systems.
– They mix and handle much like ZOE impression materials.
– The composition is similar to ZOE temporary cements.
– These products are placed over surgical sites to protect the underlying tissues.
– They are temporarily held in place by using the undercuts and embrasures of teeth.
– Surgical packs are weak materials, so they are easily removed.
– Because ZOE products have an unpleasant taste and smell, non eugenol products
have become popular.
– These products are formulated in the same manner as non-eugenol temporary cements.
E. Endodontic Sealers
– Many endodontic sealers are zinc oxide and eugenol preparations with added rosin and
barium sulfate.
ــــــــــــــــــــ►ⒹⒺⓃⓉⒶⓁ–ⓈⒸⒾⒺⓝⓒⒺ◄ــــــــــــــــــــ